odium-22 is a radioactive isotope of sodium, undergoing positron emission to 22Ne with a half-life of 2.605 years. Sodium-22 is composed of 11 protons, 11 neutrons, and 11 electrons. By measuring the concentration of this isotope, the neutron radiation dosage to the victim can be computed. Acute neutron radiation exposure (e.g., from a nuclear criticality accident) converts some of the stable 23Na in human blood plasma to 24 Sodium-23 is composed of 11 protons, 12 neutrons, and 11 electrons. Two radioactive, cosmogenic isotopes are the byproduct of cosmic ray spallation: 22Na has a half-life of 2.6 years and 24Na, a half-life of 15 hours all other isotopes have a half-life of less than one minute. Mass numbers of typical isotopes of Sodium are 23. 
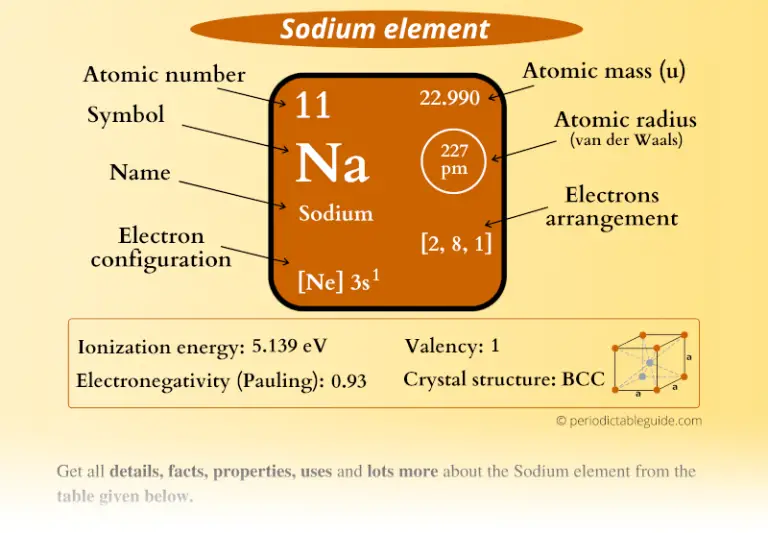
Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Sodium is a chemical element with atomic number 11 which means there are 11 protons in its nucleus.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
